An OFox in an OBox
Under the covers
Professor Alexei Demchenko, of the University of Missouri – St Louis, tells us about the whimsical names his group gives to the molecules they develop, and how they led to a fox in a box on the cover of Organic & Biomolecular Chemistry!
From the building blocks of nature to disease-battling therapeutics and vaccines, carbohydrates have had a profound impact on evolution, society, economy, and human health.
There have been numerous applications of these essential biomolecules in many areas of science and technology, mostly in the development of medicines and tools for diagnosis.
Carbohydrates with different properties are in demand for use in biology and medicine research, but they are very challenging for chemists to make. To create molecules with the right properties, my research team needs to be able to control how they come together – including their shape and orientation. To help with this, we design "leaving groups" – molecule fragments that behave consistently in leaving the molecule at a specific stage of the reaction.
We’ve developed lots of leaving groups over the years, and we have to come up with names for all of them. Some are named after their chemical formulae but not all – we’ve tried lots of different things, including naming one after the student who developed it.
The first leaving group we designed was a benzoxazole, a type of molecule based on a six-membered ring fused to a five-membered ring. We called it the SBox, after its chemical name, and a later variant was called the OBox. In this paper we present our latest variant. It’s fluorinated, so we call it the OFox.
From the art desk
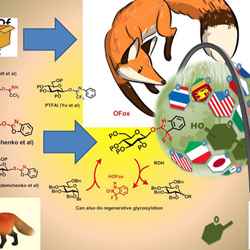
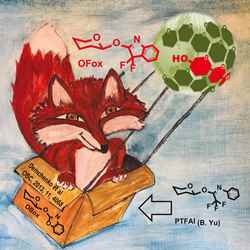
When we were invited to provide a cover, my students and I started playing around with the imagery. I already use the image of a fox in a lot of my presentations about the OFox leaving group, and at the same time we were thinking of our previous leaving group, the OBox, so one of my students, Salvo Pistorio, put the two together and came up with a first sketch of a fox in a box.
At the same time, I spoke to Fabia D’Amore-Krug, a friend of mine who is an art therapist, and showed her one of my presentation slides (see below). It featured a fox, but also my group’s logo. My group is called Glycoworld, and the logo is an image of the globe, with chemical structures on it, and flags representing the nationalities of my students.
Fabia put this idea together with the fox in a box, and sketched out the final image. Our group’s logo is now represented by a hot-air balloon, and it’s holding our two leaving groups, represented by the fox in a box.
Fabia’s initial sketch was done in pencil, then in pen, and she created her final image in beautiful watercolour.
Read the article: Swati S. Nigudkar et al, Org. Biomol. Chem., 2017, DOI: 10.1039/C6OB02230H
This image appears on the front cover of Organic & Biomolecular Chemistry, 2017, Issue 2.
Press office
- Tel:
- +44 (0) 20 7440 3351
- Email:
- Send us an email