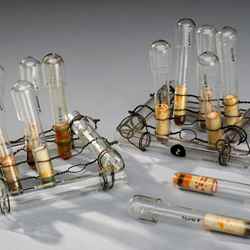
Week 12: Can you help us with #Chemglass?
Sophie Waring is Curator of Chemistry at the Science Museum, and she needs your help.
The Science Museum has a vast collection of historical glassware, and while much of it is well documented, in some cases they have very limited information about who owned it or what they used it for, which is where our members come in.
Some of the items in the collection were in use until relatively recently, and whilst we may know broadly what their purpose was, Sophie would like to know more about their practical application – what kind of experiments they would be used for, or how they worked.
If you were studying chemistry a few decades ago you might remember using some of this glassware, or you might have glassware in your lab today that looks similar, or has a similar purpose. Either way we would love to hear from you.
Some of the items are much older, and their precise use has been lost to history. Sophie is keen to hear your ideas about them. "I hope we can collaborate to generate some theories", she says. "I’m hoping to harness your user experience to improve our understanding of the chemistry collection, creating a vital resource for future historians and curators."
Each Monday for the next 12 weeks we will share a photo of a different piece of glassware – on social media and on this page – and ask for your insights, memories, and speculations.
Click on the buttons by each item below to send us your stories, and remember no contribution is too small!
Glass pestle and mortar, Europe, 1601–1700
Made from Venetian style glass, this pestle and mortar looks like it has never been used. The pestle is used to pound and grind hard substances, spices and plants in the mortar for drug preparations. The set may have been owned by a pharmacist or apothecary or could have been made for decorative purposes. Pestles and mortars are usually made from materials such as marble or bronze.
Are there any examples when a glass mortar and pestle would be useful?
Last year…
Below are the items we shared on social media in 2017. We have already received some messages from you, containing some great suggestions and interesting memories, which we plan to share at a later date. Do you have anything to add?
Press office
- Tel:
- +44 (0) 20 7440 3351
- Email:
- Send us an email