Use microscale titration to complete an acid–base neutralisation with sodium hydroxide in this class practical
In this experiment, students use a microscale titration apparatus – prepared from pipettes, a syringe and some rubber or plastic tubing – to carry out a titration, filling the ‘burette’ with hydrochloric acid and placing sodium hydroxide solution in a beaker. Students then try to calculate the exact concentration of the sodium hydroxide solution.
For this microscale technique manipulative skills are important, and students need to be capable of careful manipulation to carry the experiment out successfully. Students also need to be familiar with the concept of the mole, and capable of performing the calculations from the results of the experiment.
On such a small scale, safety issues are minimal. Similarly, the time taken to carry out a titration should be much reduced as the volumes being reacted are so small. It should be possible for a class to carry out the practical work and calculations in a one-hour session.
Equipment
Apparatus
- Graduated glass pipette, 2 cm3
- Pipette, 1 cm3, and pipette filler to fit (or a 1 cm3 plastic syringe)
- Plastic syringe, 10 cm3
- Fine-tip poly(ethene) dropping pipette (see note 6 below)
- Small lengths of rubber, plastic or silicone tubing
- Beakers, 10 cm3, x2
- Clamp stand with two bosses and clamps
Chemicals
- Dilute hydrochloric acid, 0.10 M, about 10 cm3
- Sodium hydroxide solution, approx. 0.1 M (IRRITANT), about 10 cm3
- Phenolphthalein indicator solution (HIGHLY FLAMMABLE), a few drops

Health, safety and technical notes
- Read our standard health and safety guidance.
- Wear eye protection throughout.
- Dilute hydrochloric acid, HCl(aq) – see CLEAPSS Hazcard HC047a and CLEAPSS Recipe Book RB043.
- Sodium hydroxide solution, NaOH(aq), (IRRITANT at concentration used) – see CLEAPSS Hazcard HC091a and CLEAPSS Recipe Book RB085. Students are to calculate the concentration of the sodium hydroxide solution so the bottle should not be labelled with the exact concentration.
- Phenolphthalein indicator solution (HIGHLY FLAMMABLE) – see CLEAPSS Hazcard HC032 and CLEAPSS Recipe Book RB000.
- A suitable poly(ethene) dropping pipette would be fine-tip standard, non-sterile, 3.3 cm3 capacity, such as those available from Sigma-Aldrich.
- Sargent-Welch produce eady-made microscale titration kits.
Preparing the microscale titration apparatus
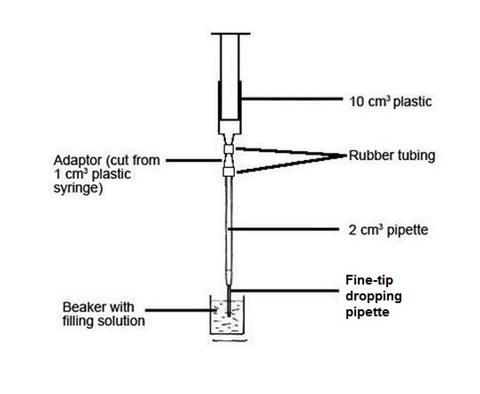
The microscale titration apparatus replaces the normal burette. To make the microscale titration apparatus, cut the tip end off a fine-tip poly(ethene) dropping pipette and push the tip carefully onto the end of a 2 cm3 graduated glass pipette. Clamp a plastic syringe, 10 cm3capacity, above the adapted pipette, as shown in the picture, and connect the two with rubber, plastic, or silicone tubing. Because the diameters of the syringe nozzle and of the top of the pipette may be quite different, two pieces of tubing, one to fit each end, will probably be needed; these can then be joined by an adaptor. A suitable adaptor can be made by cutting the lower end off a 1 cm3 plastic syringe, such that the syringe body diameter fits the wider tubing, and the syringe tip fits the narrower tubing. (See the diagram and photograph.)
It is possible for students to build their own microscale titration apparatus from supplied components, but this is likely to take the students more time than the titration itself! For that reason, it is probably preferable to prepare a class set of these in advance.
Procedure
- Clamp the microscale titration apparatus securely in position as in photograph and push the syringe plunger completely down.
- Fill the apparatus with 0.10 M hydrochloric acid as follows. Put about 5 cm3 of the acid in a 10 cm3 beaker and place the tip of the apparatus well down into the solution. Raise the syringe plunger slowly and gently, making sure no air bubbles are drawn in. Fill the pipette exactly to the zero mark. Release the plunger; the level should remain steady.
- Use the 1 cm3 pipette and pipette filler to transfer exactly 1.0 cm3 of the sodium hydroxide solution into a clean 10 cm3 beaker.
- Add one drop (no more!) of phenolphthalein indicator solution to the sodium hydroxide solution.
- Adjust the position of the microscale titration apparatus so that the tip is just below the surface of the sodium hydroxide and indicator solution in the beaker
- Titrate the acid solution into the alkali by pressing down on the syringe plunger very gently, swirling to allow each tiny addition to mix and react before adding more.
- Continue until the colour of the indicator just turns from pink to permanently colourless.
- Record the volume of hydrochloric acid added at that point.
- Repeat the titration until you get reproducible measurements – that is, the volume required is the same in successive titrations.
Calculating the concentration of sodium hydroxide solution
- The equation for the neutralisation reaction is:
HCl(aq) + NaOH(aq) → NaCl(aq) + H2O(l)
From the equation you can see that one mole of hydrochloric acid reacts with one mole of sodium hydroxide. - What was the reliable value for the volume of hydrochloric acid solution needed? Let us call this value V cm3.
- Calculate the number of moles of hydrochloric acid in this volume using the formula: V/1000 x C , where C is the concentration of the hydrochloric acid in M.
- How many moles of sodium hydroxide were therefore present in the original 1 cm3 of sodium hydroxide solution placed in the beaker?
- Now calculate how many moles of sodium hydroxide would have been present in 1000 cm3 . This is the concentration of the sodium hydroxide solution in mol dm–3.
Teaching notes
This microscale technique minimises apparatus and chemical requirements, and takes less time to perform than titration on the usual scale. Although the solutions used do present minor hazards, the use of such small quantities reduces risks from those hazards to very low levels. Students should nevertheless take all usual precautions in handling these solutions. The main risk is from misuse of the syringe or pipettes, especially if containing hazardous solutions.
The technique also makes the point that quantitative chemical experimentation does not always have to be performed on the traditional ‘bucket’ scale at school level.
Additional information
This is a resource from the Practical Chemistry project, developed by the Nuffield Foundation and the Royal Society of Chemistry. This collection of over 200 practical activities demonstrates a wide range of chemical concepts and processes. Each activity contains comprehensive information for teachers and technicians, including full technical notes and step-by-step procedures. Practical Chemistry activities accompany Practical Physics and Practical Biology.
© Nuffield Foundation and the Royal Society of Chemistry


















1 Reader's comment