The chemistry laboratory is full of hazards and it is your responsibility to work safely for your own well-being and for the well-being of others. Use this resource to understand the basic rules and procedures that should be followed to minimise any risks.
Introduction
The chemistry laboratory is full of hazards and it is your responsibility to work safely for your own well-being and for the well-being of others. Use this resource to understand the basic rules and procedures that should be followed to minimise any risks.
Contents
Laboratory rules and chemical safety
Handling glassware and personal protection
Laboratory rules and chemical safety
General principles
Hazard
A hazard is defined as the potential of a substance to cause harm. Hazards are a property of a substance and cannot be reduced, for example, concentrated sulfuric acid is corrosive, it cannot be changed (although in some situations you might be able to use dilute sulfuric acid which is less hazardous)
(For more details see ‘Hazard Warning Symbols’ for the common hazards).
Risk
Risk is how likely a substance will be harmful under the conditions it is used. Risks can be reduced by using smaller amounts of chemicals and taking precautions such as using fume cupboards.
Risk assessment
An effective risk assessment will take into account the hazards involved and who will be at risk. It also addresses the steps needed to minimise risk and will include the safe means of disposal of the substances used or made.
Basic laboratory best practice
Here are a few simple rules that form the basis of good, safe, lab practice. There might be particular procedures however that require special rules. For this reason it is vital to read any laboratory instructions thoroughly and to seek the advice of demonstrators / lab supervisors.
Dos and don’t
- Always wear eye protection (safety spectacles or goggles)
- Always wear a lab coat and make sure it is buttoned up
- Always tie back long hair
- Always wear gloves when required (dependent on risk assessment and chemicals used)
- Always be aware of hazards and risks
- Always clear up spillages immediately
- Always minimise risks by working tidily and clean up after the experiment
- Use a fume cupboard if necessary
- Never work alone or unsupervised
- Never eat or drink in the laboratory
- Never touch, sniff or taste chemicals
- Never wear open-toed shoes or sandals in the laboratory
- Never pipette liquids by mouth, always use a safety filler
- Never dispose of hazardous materials down the drain, use the appropriate waste containers provided
- Never return unused chemicals to their original containers but try to avoid taking more than you need in the first place
Handling chemicals safely
Harmful and toxic chemicals
The difference between a harmful chemical and a toxic one is a matter of degree; chemicals which are particularly harmful are classified as toxic. Harmful and toxic chemicals must always be handled in a fume cupboard with the sash (sliding front) pulled down as far as is practicable to protect yourself from splashes and explosions. It is also essential to wear protective gloves and take particular note of any special instructions about disposal and what to do in case of spillage.
Examples of harmful and toxic materials are aniline (phenylamine), bromine (most aqueous solutions and the pure liquid), chlorofrorm (trichloromethane), methanol and cyanide salts
Flammable chemicals
Many solvents and reagents used in the laboratory are highly flammable and so there should never be naked flames in places where they are being used. The heating of flammable materials should be carried out using electrically heated water baths, heating mantles or hot plates.
Examples of highly flammable solvents are diethyl ether (ethyloxyethane), petroleum ether (a mixture of hydrocarbons, not an ether), toluene (methylbenzene), acetone (propanone) and ethyl acetate (ethyl ethanoate).
Irritant chemicals
Chemicals may irritate the skin, eyes or respiratory system. Those which produce a vapour which irritates the eyes or respiratory system should always be handled in fume cupboards.
Examples of irritants are solid copper sulfate (but it does not produce an irritating vapour), styrene (phenylethene) (which does produce an irritating vapour). Acid chlorides (such as ethanoyl chloride), or thionyl chloride are both corrosive and produce irriating vapours.
Corrosive chemicals
The most commonly encountered corrosive materials are acids and alkalis although many other types of chemical fall into this category. It is essential to wear appropriate protective gloves when handling corrosive material and if there is contact with the skin it should be washed off immediately with plenty of water. It may also be necessary to seek medical attention.
Examples of corrosive materials include concentrated and even moderately dilute solutions of mineral acids (hydrochloric, nitric, sulfuric and phosphoric acids) and strong alkalis such as sodium hydroxide and potassium hydroxide.
Oxidising agents
Oxidising agents are dangerous because they can cause fires if they make contact with any combustible material, particularly if they are disposed of carelessly.
Examples of oxidising agents are solid potassium dichromate(VI), solid potassium manganate(VII), concentrated nitric acid as well as concentrated hydrogen peroxide.
Explosive reagents
Some compounds and mixtures are explosive because they are unstable, particularly if heated or when dry. Others present a risk of causing explosions because they react violently with water. When handling such chemicals it is essential to use only small amounts and protect yourself with a face mask and a safety screen.
Examples of explosion hazards are ammonium dichromate(VI) and gun powder. The alkali metals sodium and potassium present a risk of causing explosions because of their reaction with water.
Handling glassware and personal protection
Handling glassware safely
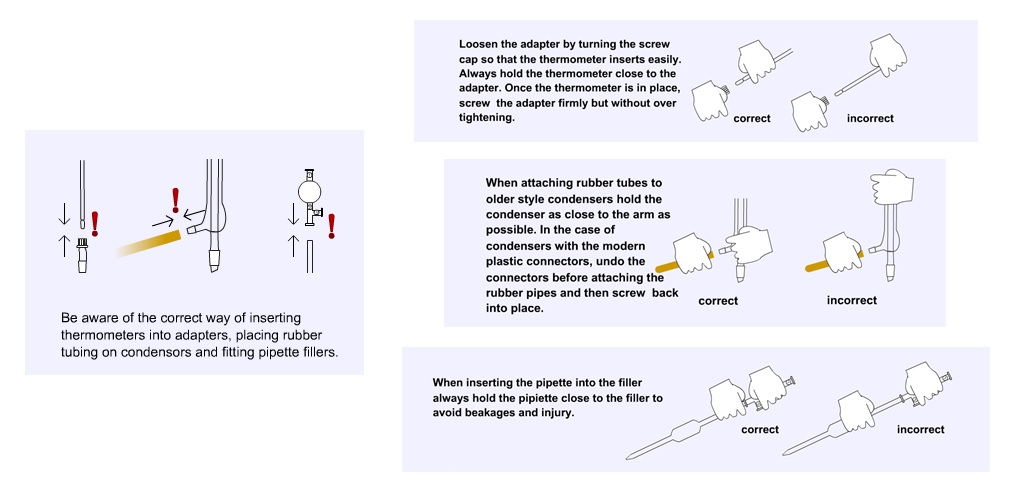
See our diagrams below which highlight some hazards and risks to be aware of when handling glassware. Careless use of glassware in the laboratory can cause many minor accidents all of which can be avoided by observing a few simple rules. If mishaps do occur always place broken glassware in the glass bin.


Personal protection
See our diagram below which highlights many of the safety rules which apply when working in university chemistry laboratories. Proper protective clothing must be worn at all times in the university chemistry laboratory to prevent contact of hazardous materials and to protect your clothing from contaminating other areas after leaving the laboratory. Please note unlike most school laboratories lab coats must always be worn and buttoned up in university laboratories.

Hazard warning symbols
Hazard warning symbols
Always make yourself aware of the hazards associated with the chemicals involved in a practical before you even start. There are two chemical hazard warning systems commonly encountered in the laboratory. The new UN-originated Globally Harmonised System (GHS) with red and black diamond hazard pictograms and the older orange and black symbols. As the older symbols are still widely encountered you should be familiar with both, please note, there is no 1:1 correlation between the old symbols and the new ones because some definitions have changed.
If you ever come across a chemical you do not know the hazard for you must assume that it is very hazardous until you find out otherwise.
GHS hazard pictograms
Some chemicals that were formerly not classified as hazardous may now carry a GHS pictogram. A GHS hazard pictogram can be accompanied by a signal word - DANGER (very hazardous), WARNING (hazardous) or no word (least hazardous) which provides some indication on how careful you need to be.
 |
 |
 |
 |
|
GHS01 Explosive materials (unstable explosives, self reactive substances and organic peroxides) |
GHS02 Flammable solids, liquids or gases. Substances which emit flammable gases on contact with water. |
GHS03 Oxidising solids liquids or gases. |
GHS04 Compressed, liquefied or dissolved gases. |
 |
 |
 |
 |
| GHS05 Corrosive to metals. |
GHS06 Acute toxicity. |
GHS07 Harmful, irritant,
and skin sensitising chemicals. |
GHS08 Damage to genetic material: mutagens, carinogens, respiratory sensitisers, hazardous substances to specific organs. |
 |
|
GHS09 Chronic hazard to aquatic environments. |
Old hazard warning symbols
 |
 |
 |
 |
|
Explosive |
Oxidising |
Flammable /extremely flammable |
Toxic/very toxic |
 |
 |
 |
 |
| Harmful/irritant | Corrosive |
Dangerous for the environment |
Radioactive |
Other sections
Return to the interactive lab primer home
Abbreviations, constants and glossary

Additional information
The interactive lab primer was developed as part of the ‘Chemistry for our future’ Royal Society of Chemistry Teacher Fellowship Scheme. With contributions from Prof. David Read (the University of Southampton), Mr David Brentnall (The University of Nottingham), Dr Sandy Wilkinson (the University of Birmingham) and Mr William Davey (The University of Sheffield).
© Royal Society of Chemistry


















No comments yet