A challenging synthesis
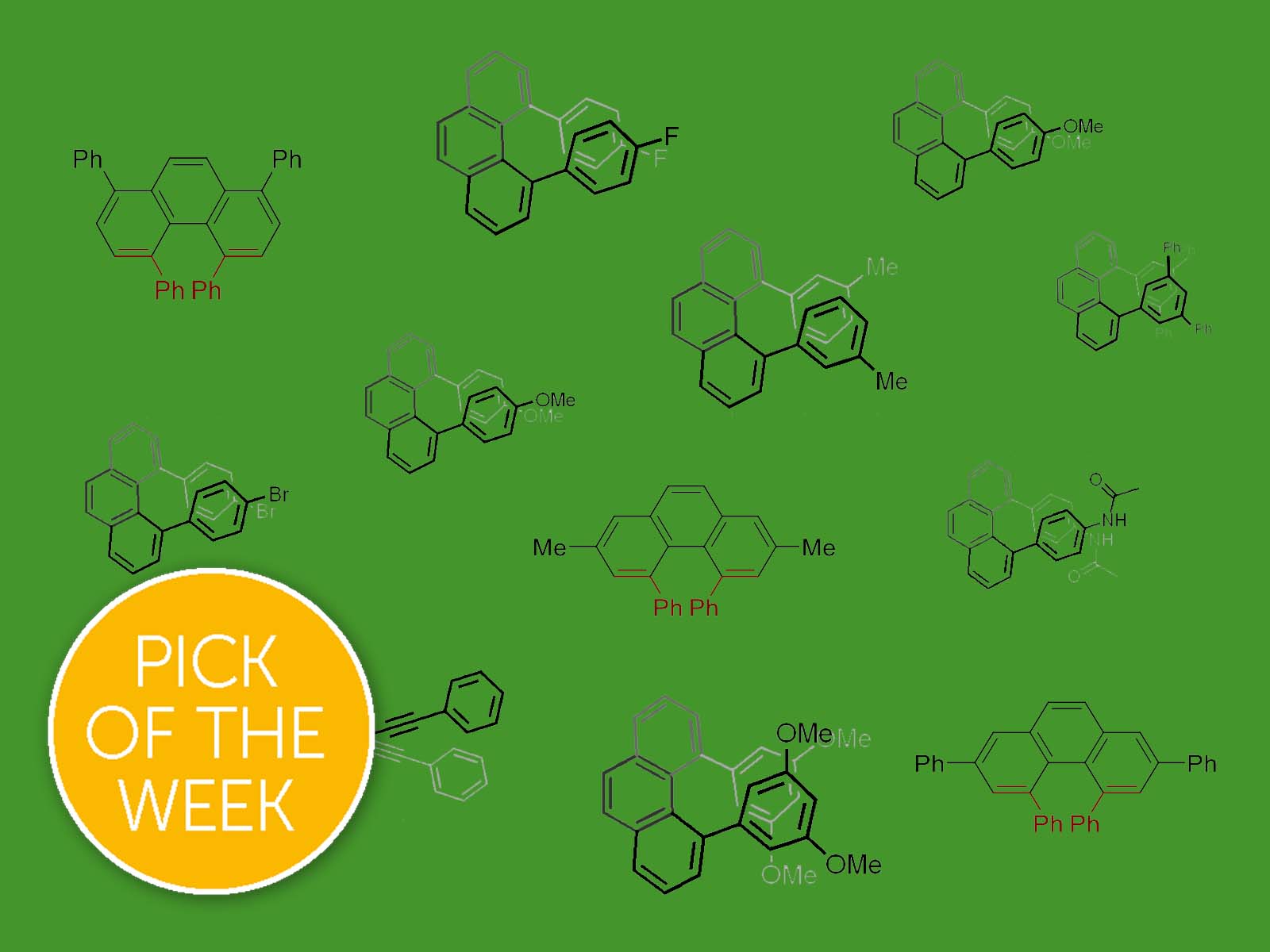
ChemSci Pick of the Week
A team of scientists in Japan have come up with a simple method to synthesise difficult-to-access twisted phenanthrene molecules.
Phenanthrene is a planar polycyclic aromatic hydrocarbon, consisting of three fused benzene rings. If we add bulky substituents into the 'bay region' of the molecule, the steric hindrance will cause the molecule to twist away from the plane. These non-planar phenanthrenes have interesting properties that open up exciting possibilities for a range of applications – from optics and electronics to anti-inflammatory and antimicrobial drugs.
Because the steric hindrance makes this type of molecule quite energetically unfavourable, it is very challenging to synthesise. Previous methods require up to five steps, and result in very low yield.
Now, Dr Kenichiro Itami and his team from Nagoya University in Japan have come up with a straightforward method. The starting materials are a benzenediacetaldehyde (a benzene ring with two aldehydes attached), and two equivalents of a phenyl alkyne. The reaction is facilitated by a Lewis acid.
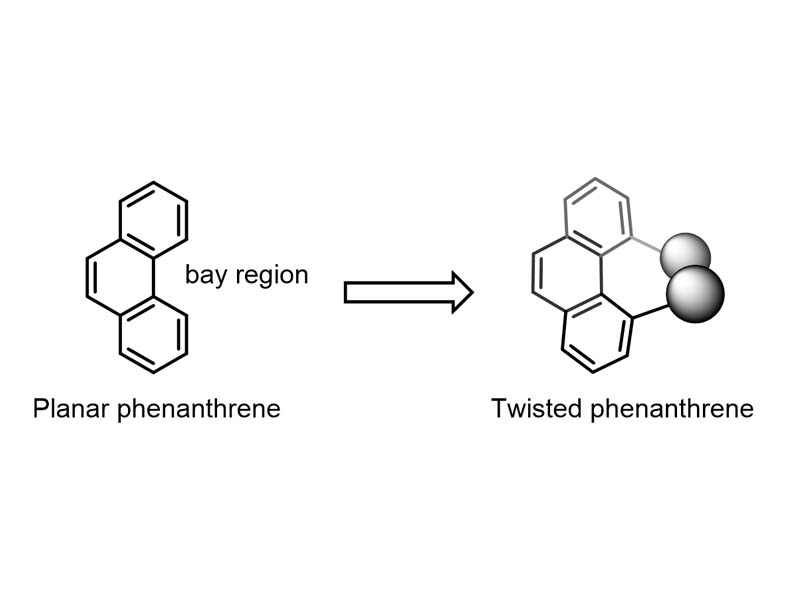
Intuitively, one might expect the phenyl groups to attach to the outside edge of the phenanthrene, away from the bay region, where there is more space. However the researchers have managed to find the perfect set of reaction conditions – including temperature, solvent, and type of Lewis Acid – to enable most phenyl groups to attach in the bay region and form the most hindered product.
Dr Itami hopes that this new simple synthesis will provide access to a host of useful molecules in future. "Due to the structural variety, good optical and electronic properties, nonplanar phenanthrenes will serve as useful building blocks for the synthesis of better organic materials, such as organic light-emitting materials and circularly polarized luminescent materials. Moreover, phenanthrene derivatives exhibit a wide range of biological activities, we hope more bioactivity of these twisted phenanthrenes will be discovered in the near future."
This article is free to read in our open access, flagship journal Chemical Science: Kenichiro Itami et al., Chem. Sci., 2019, Advance Article. DOI: 10.1039/C9SC00334G. You can access our 2019 ChemSci Picks in this article collection.