Finding a needle in a haystack
ChemSci Pick of the Week
A new infrared spectroscopy method could become a new method for blood sample analysis in the future.
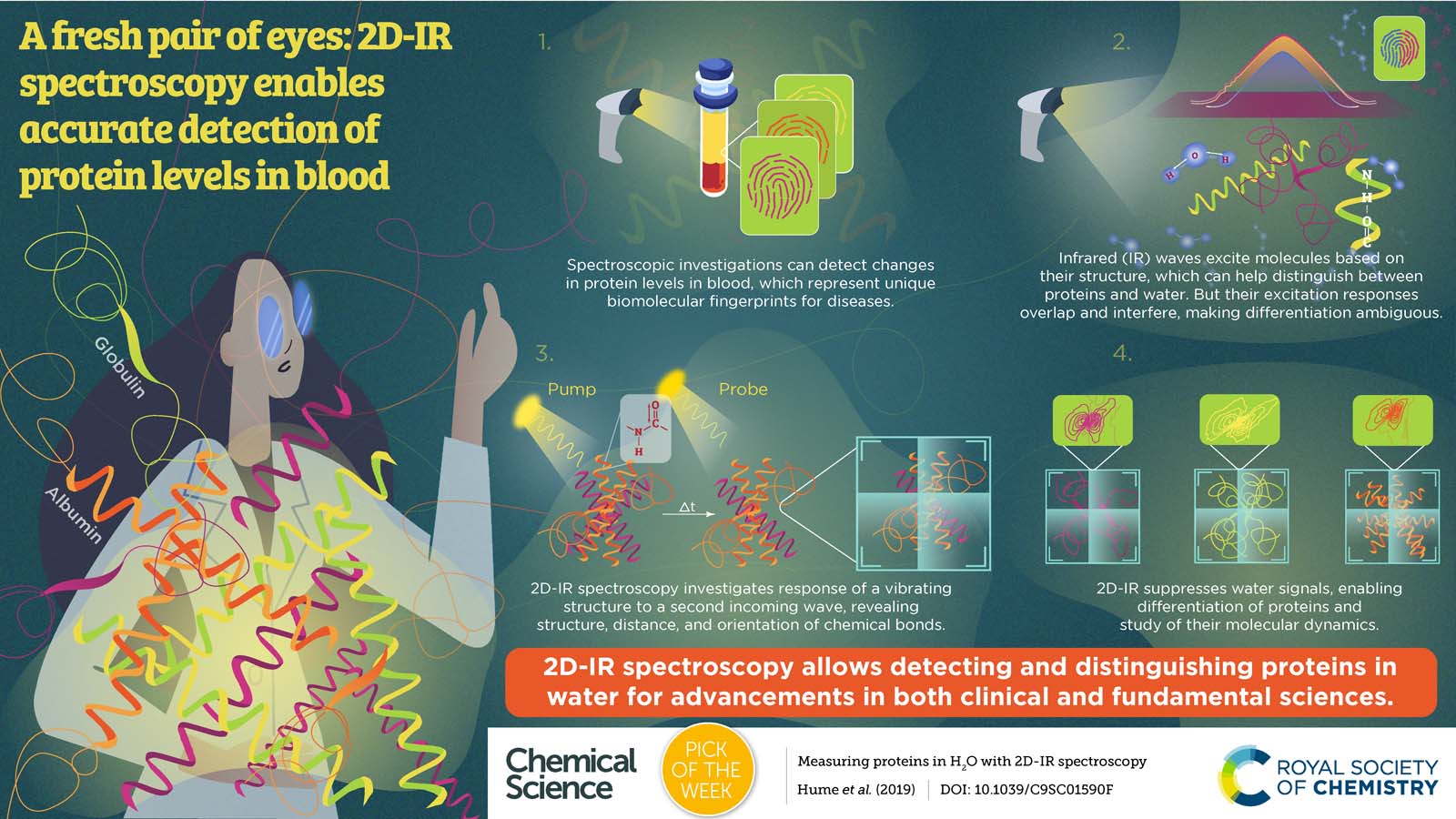
Infrared (IR) spectroscopy is a powerful tool used by chemists to identify molecules and determine their structure. It could be used to analyse the proteins in biofluids such as blood serum, in order to diagnose diseases as early as possible.
Unfortunately water absorbs IR light at the same wavelength as proteins, meaning that the signal of many proteins is blood serum is often covered up by the signal for water. Now, Professor Neil Hunt and his team from the University of York have shown that is it possible to carry out 2D IR experiments where the water does not interfere with the protein signal.
Professor Hunt says: "Imagine a protein molecule in water as being like a needle in a haystack – a small number of proteins (needles) are surrounded by a large number of water molecules (hay) that obscure our view of the proteins. The 2D-IR method makes the signal from the protein many times stronger than that from the water – the hay is effectively made transparent, so we can see the needles clearly."
"With further development work 2D-IR could become a new method for blood sample analysis. An alternative future direction could see 2D-IR being used in drug design projects. 2D-IR is a powerful tool for studying protein-drug binding but currently scientists need to replace H2O with expensive D2O to use 2D-IR. Our method removes that need, making the application of information-rich 2D-IR spectroscopy to protein-drug systems much more economical, straightforward and so accessible."
This article is free to read in our open access, flagship journal Chemical Science: Neil Hunt et al., Chem. Sci., 2019, Advance Article. DOI: 10.1039/C9SC01590F. You can access our 2019 ChemSci Picks in this article collection.
Read more like this